Solid Phase Reactor - Advanced Solution for Pharmaceutical & Chemical Processing
Xi'an Bioland Instrument Co., Ltd. makes high-performance solid phase reactor systems designed for making drugs, peptides, and chemical intermediates. High-borosilicate glass construction that can withstand temperatures up to 200°C, built-in PTFE sandcore filtration (G2-G3 grade, 15-80μm pore size), and precise PID temperature control that is accurate to within 0.1°C. Offering explosion-proof certifications (CE, ISO, UL, SGS, ATEX), automatic PLC control choices, and the ability to combine ultrasonic crystallization, vacuum distillation, and filtration processes in a modular way, our systems work exceptionally well in complex pharmaceutical settings. Operating temperature range: -80°C to 250°C. Adhering to GMP/FDA standards guarantees clean output for peptide APIs and antimicrobial study uses.

Product Specifications
1.The whole stainless steel column, mobile framework, 5-7 or more covers of the reaction kettle, with full set of glass parts for reflux, adding liquid, temperature measurement, and so on.2. Use G3.3 high borosilicate glass, with good chemial and physical properties.

3. Alloy steel mechanical seal, PTFE connector, to maintain a high precision seal in the working condition.
4. With PT100 sensor probe, high precision temperature measurement, small errors, high efficiency.
5. AC reducer motor, strong torsion, no noise.
6. PTFE discharge value, moveable interface, discharging completely and quickly.
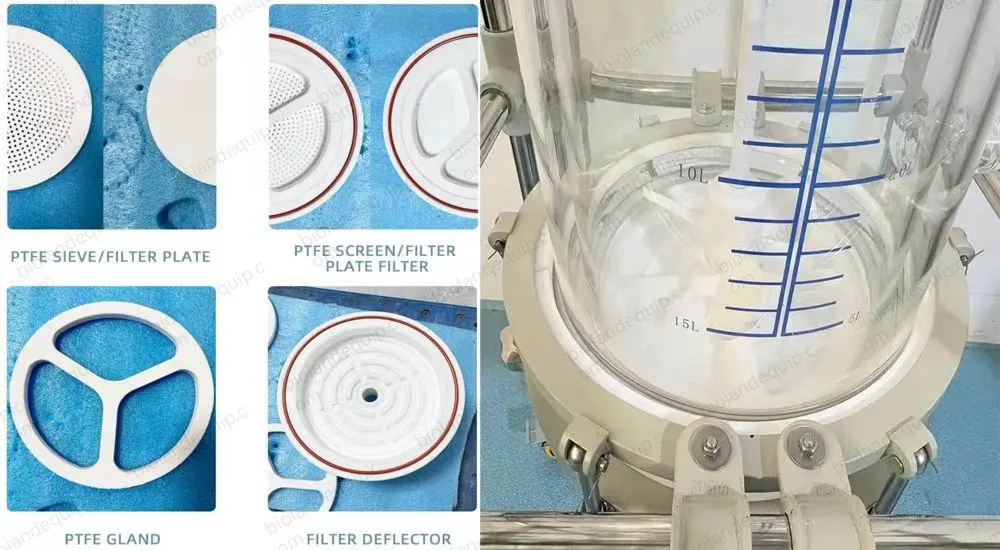
7. Our company can design special kettle boday structure according to customer requirements(such as PH meter, sand core filter etc)
8. All the reaction kettle can be made of explosion-proof device according to the customer request.(The explosion-proof device refers to the ex-proof motor + ex-proof control box + ex-proof temperature field display instrument).

Why Choose Our Solid Phase Reactor Systems?
Industry-Leading Quality & Compliance
The gear you get is made to meet foreign standards. The reactors we sell are certified by CE, ISO, UL, SGS, and ATEX. From planning to making, every unit meets GMP and FDA standards. When it comes to medicine uses, you can trust the quality.
Advanced Safety Features
When you do your work, safety comes first. Our high-strength pressure-bearing buildings and electricity systems that can't explode will keep your team safe. Human mistake is less likely with PLC automatic control. German-made seals and variable-frequency spinning make sure that the machine will work steadily for a long time with little upkeep.

Complete Process Visualization
You can see what's going on inside. You can see crystallization, filter, and reaction dynamics happen in real time through the clear high borosilicate glass body. Being able to see what's going on helps you improve processes and find problems early.
Exceptional Chemical Resistance
Your chemicals will not damage our tools. The body of the reactor can handle acids, alkalis, and organic fluids without affecting the result. TFA, piperidine, and DCM are strong chemicals that don't bother parts that are wrapped with PTFE or have surfaces that have been passivated.
Modular Flexibility
Set up the setup the way you want. We use one platform for ultrasonic temperature-controlled reaction, crystallization, filtering, distillation, and vacuum operation. You only get what your business needs, nothing more or less.
Custom Solutions at Factory Prices
It's better for you to buy things that are made directly. We can fully customize OEM/ODM orders, including sizes, materials, types of stirring, and control systems that are made to fit your needs. Standard models ship in 5 to 7 days, while customized units ship in 30 business days.

Professional Support Throughout
Your success is our priority. A dedicated specialist tracks your project weekly with photos and videos. Before shipment, you receive detailed inspection materials or can schedule a Factory Acceptance Test at our facility.
Technical Parameters
|
MODEL
|
BL-FR10L
|
BL-FR20L
|
BL-FR30L
|
BL-FR50L
|
BL-FR100L
|
|
Material capacity
|
10L
|
20L
|
30L
|
50L
|
100L
|
|
Jacket capacity
|
≈2L
|
≈5L
|
≈8L
|
≈15L
|
≈20L
|
|
Receiving flask
|
5L
|
10L
|
10L
|
20L
|
20L
|
|
Reactor body and cover
|
Apply vacuum grease to the connection surface between the upper part of the kettle body and the kettle cover.
|
|
Variable-frequency governor
|
Digital temperature and rotate speed
|
|
Rotate speed
|
0-300r/min
|
|
Seal
|
Self-lubricating mechanical seal
|
|
Filter base plate
|
PTFE filter sand core
|
|
Bore diameter of sand core
|
1-200μm (Optional)
|
|
Motor power
|
120W/1:3
|
200W/1:3
|
|
Power of explosion-proof motor
|
180W
|
370W
|
370W/1:3
|
|
Voltage
|
220V/50Hz which can be customized according to customer requirements
|
Working Principle
The solid phase reactor operates through controlled thermal and chemical processes while maintaining materials in solid or semi-solid states.
In pharmaceutical synthesis, the reactor tank holds resin beads supported by polymers that are mixed with a liquid. The jacketed design heats or cools the response zone precisely across the whole area. With variable-speed shaking, the glue is spread out evenly without too much shear, which could damage the structure of the bead.
A lot of chemicals run through the bottom inlet valve during peptide synthesis. The stirring method makes sure that the active sites and coupling agents are in full touch with each other. Controlling the temperature stops side reactions and keeps exothermic reactions under control. The built-in PTFE filter plate keeps the resin beads in place after each coupling step while letting the solvent drain quickly through the bottom exit.
The vacuum system gets rid of reaction by-products and water, which speeds up the reaction process. Using inert gas to purge keeps the conditions dry, which is important for sensitive chemicals.
For crystallization processes, the reactor exactly sets the rate at which the material cools. You can see how the crystals form and grow through the clear body. Ultrasonic units, which are extra, help crystals grow evenly and control the range of sizes they grow to.
All the factors—temperature, pressure level, stirring speed, and process timing—are coordinated by the PLC control system. This makes sure that the results are the same from batch to batch.

Application Fields
Pharmaceutical API Manufacturing
Your work on developing peptide drugs needs to be as pure as possible. Our reactors are very good at making GLP-1 agonists, thymic peptides, and antibacterial chemicals in the solid phase. The system keeps strict temperature cycling over long runs and achieves connection efficiencies of 99% or more at each step.
Chemical Intermediate Production
Feel confident as you make high-value fillers. In one tank, the reactor does multiple steps of synthesis, crystallization, and cleaning. Chemical compatibility makes sure that your chemicals and reagents won't damage your equipment.
Bioengineering Research
You need flexible tools for everything from immobilizing enzymes to bioconjugation. Our reactors can follow a variety of routines while keeping the surroundings clean and under tight control.
New Materials Development
Create new plastics and materials that do useful things. The flexible design and wide temperature range make it possible to use experimental methods from the first screening to trials on a larger scale.
Oligonucleotide Synthesis
For making DNA and RNA medicines, our reactors keep the conditions completely dry. During synthesis, the purity of the nucleotides is maintained by quick, automatic solvent changes and precise control of the oxidation step.
Combinatorial Chemistry
Screen compound libraries efficiently. The reactor maintains uniform resin suspension across varying densities without clogging filtration media, accelerating your discovery timelines.
Quality Certification
Xi'an Bioland Instrument has a lot of foreign approvals, including:
- CE Certification: Meeting European health and safety standards
- ISO 9001 is a certification for a quality management system.
- UL Certification: Safety rules for North America
- SGS Verification: An independent check of quality
- Explosion-proof device guidelines from ATEX and IEC
GMP rules are used to make all of our reactor systems. The materials, bonding methods, and control systems used in pharmaceutical production meet the standards. There are material certificates, pressure test results, and validation procedures in documentation packages.
During production, our plant keeps a close eye on quality. Before it is shipped, each reactor is tested for hydraulic pressure, leaks, and functionality.
FAQ
Q: What's the lead time for customized reactors?
Customized units typically require 30 business days from order confirmation. Standard configurations ship within 5-7 business days. We provide weekly progress updates with photos during production.
Q: Can you modify existing designs for specific solvents?
Absolutely. We engineer material selection and seal compatibility based on your chemical environment. Provide your solvent list and operating parameters for our technical assessment.
Q: How do you ensure temperature uniformity?
Our jacket design provides even thermal distribution. PID control with RTD sensors maintains ±0.1°C accuracy. For critical applications, we can add multiple temperature monitoring points.
Q: What maintenance does the reactor require?
Routine maintenance involves seal inspection and filter cleaning. We use German-imported seals for extended service life. Your maintenance intervals depend on operating conditions, but typically occur every 6-12 months.
Q: Do you offer installation and training?
Yes. We provide detailed operation manuals, installation guidance, and can arrange on-site training. Remote video support helps your team master the system quickly.
Q: Can the reactor integrate with our existing equipment?
Our modular design facilitates integration. We'll work with your process flow diagrams to ensure compatibility with your upstream and downstream equipment.
Q: What warranty coverage do you provide?
All reactors include a one-year quality warranty with lifetime maintenance support. We maintain spare parts inventory for rapid replacement if needed.
Q: How do I verify the reactor meets our requirements before shipping?
Review detailed photos and videos we provide upon completion, or schedule a Factory Acceptance Test at our Xi'an facility. We don't ship until you approve.
Contact Us
Ready to enhance your pharmaceutical or chemical processes with a solid phase reactor? Reach out to discuss your application requirements. Email us at info@biolandequip.com for technical consultation and quotations.

















